Management of Adenomyosis: A Single Centre Retrospective Study of Hysterectomy and LNG-IUS treated patients
Management of Adenomyosis: A Single Centre Retrospective Study of Hysterectomy and LNG-IUS treated patients
Kiran Mehndiratta1*, Shailesh Perdalkar2, Shubha Shankari3, Shubha Dadhich4, Rohit Ravindra Dusane5, Mary Boban6
1,3,4,6 - Department of Obstetrics and Gynaecology, NMC Specialty Hospital, Abu Dhabi, United Arab Emirates.
2,5- Clinical Research Department, NMC Healthcare, Abu Dhabi, United Arab Emirates.
*Correspondence to: Kiran Mehndiratta, Consultant Obstetrics and Gynaecology, NMC Specialty Hospital, Abu Dhabi, United Arab Emirates.
Copyright
© 2025 Kiran Mehndiratta. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 25 June 2025
Published: 07 July 2025
Management of Adenomyosis: A Single Centre Retrospective Study of Hysterectomy and LNG-IUS treated patients
Introduction
Adenomyosis (AD) is a common benign gynaecologic condition in which the uterus is enlarged due to myometrial invasion of endometrial glands and stroma. AD can be asymptomatic or may induce menorrhagia, dysmenorrhea, abnormal uterine bleeding, pelvic pain, hypermenorrhoea, dyspareunia, subfertility or infertility and eventually affecting the quality of life of the women[1–3]. The pathophysiology of AD is not clearly understood, but several theories have been proposed over the years [4–6]. Age, parity, and previous history of uterine surgery increase the risk of developing AD [Schrager S et al 2022]. The diagnosis of AD is challenging due to the overlapping symptoms with other gynaecological conditions such as endometriosis, uterine fibroids and leiomyomas.
Earlier AD could only be diagnosed by histopathological examinations in patients undergoing hysterectomy, thus the diagnosis was restricted to patients who did not wish to preserve their fertility. However, the advancement in imaging techniques such as transvaginal ultrasound (US) and Magnetic Resonance Imaging (MRI), have increased the scope of AD diagnosis in younger women as well [7]. The quest for a reliable biomarker for the diagnosis of AD is ongoing and currently, there are no clinically reliable biomarkers available for the same. However, studies investigating the use of CA 125[8], Cytokines [9], miRNA [10], proteomics and metabolomics [11, 12] for the diagnosis or differential diagnosis of AD have gradually increased. In future, AD diagnosis may be benefited from the integration of biomarkers along with imaging techniques.
Hysterectomy is the conventional treatment for symptomatic patients [13] as at present, there are no international guidelines on the clinical and surgical management of AD. Although hysterectomy is an effective treatment option for AD in providing long-term relief from symptoms and preventing recurrence, it often conflicts with patients' desire to preserve their fertility [14]. It is further compounded by surgical complications, short-term morbidities, and mortality [15]. Minimally invasive techniques like uterine artery embolization, laparoscopic myometrial resection/ electrocoagulation, endo-myometrial ablation, MRI-guided focused US (MRgFUS), high intensity focused ultrasound (HIFU), etc. are also used for symptomatic management of AD. Considering the lack of high-quality data, more evidence on these treatment options is warranted [16, 17].
The pharmacological management of AD includes NSAIDS for pain relief, hormonal therapies such as gonadotropin-releasing hormone analogues (GnRH-a), oral contraceptives, high-dose progestins, and hormone receptor modulators. According to the National Institute for Health and Care Excellence guidelines, the first-line treatment for AD should be an intrauterine hormone-releasing device [16, 18]. Levonorgestrel-releasing intrauterine system (LN-IUS) is a surgically implanted medical device that releases levonorgestrel locally into the uterus to treat the symptoms of AD temporarily [3]. LNG-IUS has been successfully used to treat AD to reduce blood loss and pain during menstruation by reducing the thickness of the myometrial junctional zone and total uterine volume. Low side effects, low serum hormonal levels, high local concentration around the site of implantation etc., are some of the advantages of LNG- IUS when compared to other oral treatments [19]. LNG-IUS offers a viable option for women who wish to preserve their fertility while managing AD [20]. Recently, there has been an increase in the number of studies that explore the benefits of LNG-IUS in AD either as a monotherapy or in combination with conservative surgeries/minimally invasive treatments/ other medical therapies. However, the studies investigating the duration and factors associated with the retention of the LING-IUS device are limited.
In this study, we intend to evaluate the characteristics of AD patients managed with either hysterectomy or LNG-IUS and explore the factors associated with the retention of LNG-IUS. Additionally, we studied the effect of LNG-IUS on pain levels in AD patients.
Materials and Methods
Study design:
A retrospective study was conducted at the Obstetrics and Gynaecology Department of NMC Specialty Hospital, Abu Dhabi. Patients aged 18 and above with confirmed diagnosis of AD between January 2020 and December 2022 were included in the study. AD with or without endometriosis and or fibroids, treated by hysterectomy or LNG-IUS were included and AD patients treated by other treatment modalities were excluded. The demographic and clinical characteristics of the patients were collected from medical records.
The diagnosis of AD was done based on symptoms of menorrhagia, menometrorrhagia, dysmenorrhea, dyspareunia, chronic pelvic pain, genitourinary or irritable bowel symptoms, supplemented with transvaginal US. Additionally, few patients had confirmed AD diagnosis by MRI.
The patients enrolled in the study were divided into two groups based on their treatment options. Group I - AD patients treated with Hysterectomy and Group II – AD patients treated or managed with LNG-IUS. Group II was further categorized based on the status of Intrauterine Device (IUD); LNG-IUS at 6 months post insertion into Group II a- LNG-IUS retained and Group II b- LNG-IUS expelled (Figure 1). For Group I, only the baseline data was collected and for Group II Baseline, 1-month (1M), 3-month (3M) and 6-month (6M) follow-up data post-insertion of LNG IUS were collected.
Statistical analysis:
Write about the analysis. Variables with P-values below 0.05 in the univariate analysis were further examined in the multivariate analysis. All statistical analyses were performed using SPSS, version xxxx. p-values less than 0.05 were considered statistically significant.
Ethics approval:
This study was approved by the NMC Regional Research Ethics Committee (NMC/RREC/AUH/2023/0013).
Tables:
Table 1: Characteristics of patients in Group I and Group II
|
Variable |
LNG IUS |
Hysterectomy |
p value |
|
|
Age |
43.11 ± 5.45 |
45.49 ± 4.47 |
< 0.05 |
|
|
BMI |
28.14 ± 4.76 |
28.17 ± 5.08 |
0.8153 |
|
|
Parity |
0 |
0 |
5.00 (8.77) |
< 0.05 |
|
1 |
14.00 (18.92) |
14.00 (24.56) |
||
|
2 |
37.00 (50.00) |
28.00 (49.12) |
||
|
>=3 |
23.00 (31.08) |
10.00 (17.54) |
||
|
Coexisting Uterine disease (Endometriosis and or Fibroids) |
NO |
19.00 (25.68) |
6.00 (10.53) |
< 0.05 |
|
YES |
55.00 (74.32) |
51.00 (89.47) |
||
|
History of C-section SX |
NO |
31.00 (41.89) |
44.00 (77.19) |
< 0.05 |
|
YES |
43.00 (58.11) |
13.00 (22.81) |
||
|
History of D&C |
NO |
54.00 (72.97) |
45.00 (78.95) |
0.430 |
|
YES |
20.00 (27.03) |
12.00 (21.05) |
||
|
NRS pain score |
2 |
19.00 (25.68) |
15.00 (26.32) |
< 0.05 |
|
4 |
50.00 (67.57) |
30.00 (52.63) |
||
|
6 |
5.00 (6.76) |
12.00 (21.05) |
||
|
Haemoglobin |
≤8 |
11.00 (14.86) |
18.00 (31.58) |
0.072 |
|
8-10 |
19.00 (25.68) |
11.00 (19.30) |
||
|
>10 |
44.00 (59.46) |
28.00 (49.12) |
||
|
PBAC |
≤80 |
9.00 (12.16) |
7.00 (12.28) |
0.180 |
|
80-120 |
46.00 (62.16) |
27.00 (47.37) |
||
|
>120 |
19.00 (25.68) |
23.00 (40.35) |
||
|
Menorrhagia |
NO |
7.00 (9.46) |
10.00 (17.54) |
0.172 |
|
YES |
67.00 (90.54) |
47.00 (82.46) |
||
|
Irregular Bleeding |
NO |
3.00 (4.05) |
10.00 (17.54) |
< 0.05 |
|
YES |
71.00 (95.95) |
47.00 (82.46) |
||
|
Dysmenorrhea |
NO |
26.00 (35.10) |
18.00 (31.58) |
0.669 |
|
YES |
48.00 (64.86) |
39.00 (68.42) |
||
|
Dyspareunia |
NO |
37.00 (50.00) |
29.00 (50.88) |
0.921 |
|
YES |
37.00 (50.00) |
28.00 (49.12) |
||
Table 2: Baseline characteristic of the Group II patients
|
Variable |
Outcome |
||||||||
|
Yes |
No |
P value |
|||||||
|
BMI |
28.32 ± 4.31 |
27.77 ± 5.69 |
0.726 |
||||||
|
Age |
42.45 ± 5.70 |
44.57 ± 4.65 |
0.110 |
||||||
|
Parity |
1 |
12.00 (0.24) |
2.00 (0.09) |
0.268 |
|
||||
|
2 |
23.00 (0.45) |
14.00 (0.61) |
|
||||||
|
>=3 |
16.00 (0.31) |
7.00 (0.30) |
|
||||||
|
Coexisting Uterine disease (Endometriosis and or Fibroids) |
NO |
16.00 (0.31) |
3.00 (0.13) |
0.095 |
|
||||
|
YES |
35.00 (0.69) |
20.00 (0.87) |
|
||||||
|
History of C-section SX |
NO |
23.00 (0.45) |
8.00 (0.35) |
0.405 |
|
||||
|
YES |
28.00 (0.55) |
15.00 (0.65) |
|
||||||
|
History of D&C |
NO |
40.00 (0.78) |
14.00 (0.61) |
0.115 |
|
||||
|
YES |
11.00 (0.22) |
9.00 (0.39) |
|
||||||
|
NRS pain score |
2 |
9.00 (0.18) |
10.00 (0.43) |
0.062 |
|
||||
|
4 |
38.00 (0.75) |
12.00 (0.52) |
|
||||||
|
6 |
4.00 (0.08) |
1.00 (0.04) |
|
||||||
|
Haemoglobin |
≤8 |
5.00 (0.10) |
6.00 (0.26) |
0.086 |
|
||||
|
8-10 |
16.00 (0.31) |
3.00 (0.13) |
|
||||||
|
>10 |
30.00 (0.59) |
14.00 (0.61) |
|
||||||
|
PBAC |
≤80 |
8.00 (0.16) |
1.00 (0.04) |
0.373 |
|
||||
|
80-120 |
30.00 (0.59) |
16.00 (0.70) |
|
||||||
|
>120 |
13.00 (0.25) |
6.00 (0.26) |
|
||||||
|
Menorrhagia |
NO |
6.00 (0.12) |
1.00 (0.04) |
0.424 |
|
||||
|
YES |
45.00 (0.88) |
22.00 (0.96) |
|
||||||
|
Irregular Bleeding |
NO |
3.00 (0.06) |
0.00 (0.00) |
0.548 |
|
||||
|
YES |
48.00 (0.94) |
23.00 (1.00) |
|
||||||
|
Dysmenorrhea |
NO |
12.00 (0.24) |
14.00 (0.61) |
< 0.05 |
|
||||
|
YES |
39.00 (0.76) |
9.00 (0.39) |
|
||||||
|
Dyspareunia |
NO |
20.00 (0.39) |
17.00 (0.74) |
< 0.05 |
|
||||
|
YES |
31.00 (0.61) |
6.00 (0.26) |
|
||||||
Table 3: Results Univariate and Multivariate regression analysis to identify the baseline predictors of LNG-IUS retention
|
|
Univariate regression ODDS ratio (CI 95%) |
p value |
Multivariate regression ODDS ratio (CI 95%) |
p value |
|
Dysmenorrhea |
5.06 (1.75 - 14.57) |
< 0.05 |
5.08 (1.65 - 15.66) |
< 0.05 |
|
Dyspareunia |
4.39 (1.48 - 13.03) |
< 0.05 |
4.42 (1.38 - 14.12) |
< 0.05 |
Table 4: Pain score in Group II patients
|
|
Baseline |
1 Month |
3 Month |
6 Month |
|
NRS Pain score |
3.62 ±1.08 |
2.00 ± 0.66) |
1.07 ± 1.07 |
0.78 ± 0.99) |
Figure 1
Results
Baseline Characteristics:
Out of 131 patients diagnosed with AD, between January 2020 and December 2022, 43.51% underwent hysterectomy, while 56.49% received LNG-IUS treatment at our facility. (Figure 1). The demographic characteristics of patients in Group I and Group II varied significantly. There was a significant difference in the mean age (p < 0.05), parity (p < 0.05), presence of co- existing uterine disease (p < 0.05) and history of caesarean surgery (C-section) (p < 0.05) in patients from the two groups (Table 1). Additionally, the pain score and irregular bleeding at baseline also varied significantly (p< 0.05). However, there was no significant difference between the two group’s BMI, history of dilation and curettage (D&C), pictorial blood loss assessment chart (PBAC) score, menorrhagia, dysmenorrhea, or dyspareunia at baseline.
Among the 74 patients treated with LNG-IUS (total follow-up 6 months) the mean duration of LNG-IUS retention was 4.32 ±2.39 months (Mean ± SD). The rate of LNG-IUS retention at 6 months was 68.92%.
In a sub-group analysis of patients treated with LNG-IUS, the patients who retained LNG-IUS for at least six months (Group IIa) were compared with the patients who expelled LNG-US within six months of insertion (Group IIb). Out of the 74 patients treated with LNG-IUS, 51 (68.92%) patients retained LNG-IUS (Group IIa), and 23 (31.08 %) expelled LNG-IUS (Group IIb). The demographic characteristics of patients in both groups did not vary significantly. There was no significant difference in the mean BMI and mean age of the patients in Groups IIa and IIb. The mean age in Group IIa was slightly lower when compared to Group IIb (42.45 ± 5.70 vs 44.57 ± 4.65), on the other hand, the mean BMI was higher in Group IIa in comparison to Group IIb (28.32 ± 4.31 vs 27.77 ± 5.69). The number of patients with coexisting uterine-related disease, a history of C-section, and a history of D&C were slightly higher in Group IIa. At baseline, the patients with Hb <8 were 10% and 26% in Group IIa and IIb respectively. The proportion of patients with Hb of 8-10 was high in Group IIa (31%) compared to Group IIb (13%). On the other hand, the proportion of patients with baseline PBAC scores of 80-120 were lower in Group IIa than in Group IIb (59% vs 70%). The percentage of patients experiencing baseline menorrhagia and irregular bleeding was low in Group IIa compared to Group IIb (Table 2). Additionally, there was a significant difference in the percentage of patients experiencing dysmenorrhea and dyspareunia at baseline in Group IIa and Group IIb (dysmenorrhea- 76% vs 39%, dyspareunia- 61% vs 26%).
Out of the 23 patients in group IIb who did not retain LNG-IUS, 13 patients (56.52%) expelled the device and in 10 patients (43.48%) the device was removed.
Predictors of successful LNG-IUS retention at 6M:
We conducted a univariate analysis to identify baseline patient characteristics that could predict the retention success of LNG-IUS. In this analysis, only dysmenorrhea and dyspareunia demonstrated statistically significant association (p < 0.05), exhibiting odds ratios of 5.06 (95% CI, 1.75 - 14.57) and 4.39 (95% CI, 1.48 - 13.03), respectively (Table 2, Figure 2). The findings were corroborated by multivariate analysis, which also yielded odds ratios of 5.08 (95% CI, 1.65 - 15.66) and 4.42 (95% CI, 1.38 - 14.12).
Figure 2: Graphical representation of the factors associated with LNG-IUS retention in AD patients treated with LNG-IUS
Pain score comparison in LNG-IUS treated patients:
The pain levels in patients in group IIa were evaluated using the Numerical Rating Scale (rang e 0- 9) at various time points: baseline, 1M, 3M, and 6M post LNG-IUS insertion. At baseline, the average pain score was 3.62 (SD ±1.08). Subsequently, the pain scores showed significant reductions to 2.00 (SD±0.66) at 1M, 1.07 (SD±1.07) at 3M, and 0.78 (SD±0.99) at 6M. However, there was no statistically significant difference in pain scores between 3M and 6M (table 4).
Discussion
AD has historically been associated with older women, often due to higher parity. However, advancements in technology and non-invasive diagnostic techniques are leading to an increased diagnosis of AD in younger women. Hysterectomy remains the gold standard treatment for AD, with benefits such as no disease recurrence, successful alleviation of pain and resolution of abnormal uterine bleeding. However, it is not recommended for individuals who wish to preserve their fertility conservative treatments are preferred in such cases [21].
Advances in research have led to the development of pharmacological treatments and minimally invasive procedures for the management AD. However, further evidence in this regard is needed to establish safety and efficacy of these treatment modalities. LNG-IUS, initially used as a reversible contraceptive, has demonstrated non-contraceptive gynaecological health benefits and was found to be effective in the management of the symptoms of AD for over a decade [13, 22, 23].
In this retrospective study, we compared the baseline characteristics of the AD patients who underwent hysterectomy with patients who were treated with LNG-US. We also identified the factors associated with the LNG IUS retention. Our study found that patients treated with LNG- IUS were significantly younger when compared to those who underwent hysterectomy (LNG- IUS vs HYT, 43.11± 5.45 vs 45.49 ± 4.47). However, we did not find significant difference in the mean BMI among the two treatment groups (LNG-IUS vs HYT 28.14 ± 4.76 vs 28.17 ± 5.08). The proportion of women with two or more prior deliveries were higher in the LNG IUS treatment group (LNG-IUS vs HYT 81.08 vs 66.66%). The LNG-IUS group had a greater number of patients with the previous history of C-section, D&C, baseline irregular bleeding and higher baseline pain score when compared to the hysterectomy group. Supporting our finding, a study by Ota I et al also showed that frequency of previous C-section was higher (52.4%) in AD patients treated with LNG-IUS [19]. On the other hand, frequency of uterine related disease was higher in Hysterectomy group in our study.
Out of the 74 patients treated with LNG-IUS, 51 (68.92%) retained the device at 6 months, which slightly higher than the retention rates in the previous studies reported in the literature [20, 24, 25]. Chen et al. reported a cumulative LNG-IUS retention rate of 57.49% (36 months follow-up) among 207 AD patients [24], and Li et al. reported a cumulative retention rate of LNG-IUS as 56.2% (60 months follow up) [20]. However, it is important to note that the follow-up periods in these studies were longer. The mean duration of LNG-IUS retention was found to be 4.32 ±2.39 months in our study, however some recent studies show that the mean duration of LNG-IUS to be 58.35 ± 15.98 months and 11.58 ± 6.22 months [25] . The lower duration of LNG-IUS retention in our study may be due to that fact that it was a retrospective study and eligible patients’ data up to 6 six months were only collected for this study.
The pain scores reduced significantly at 1M, 3M and 6M when compared to the baseline value and the preceding period, which agrees with the result from previous studies [20, 26, 27]. A meta-analysis by Abbas.et.al. reported a similar observation, the overall effect of LNG-IUS significantly reduced the pain score at 12, 24 and 36 months and the standard mean difference (SMD) were found to be −3.87 (95% CI −5.51 to −2.23) at 12 months, −5.56 (95% CI −9.80 to −1.32) at 24 months and −3.81 (95% CI −4.27 to −3.36) at 36 months after LNG IUS insertion [16].
A study by Shaaban.et.al., showed that LNG-IUS decreased the pain associated with AD from 6.23±0.67 to 1.68±1.25. It also conveyed that this effect may be secondary to the reduction in the uterine volume and the increase in resistance to blood flow[26].
In Group II, there was no significant association between the clinical characteristics of patients who retained LNG-IUS (Group IIa) and those who expelled LNG-IUS (Group IIb), except for the presence of baseline dysmenorrhea (39% vs. 9%) and dyspareunia (31% vs. 6%), which were significantly higher in Group IIa compared to Group IIb. Only a few studies have explored the factors associated with LNG-IUS retention in AD patients [28]. The current study found that the presence of baseline dysmenorrhea (OR 5.08, 95% CI 1.65 - 15.66) and dyspareunia (OR 4.42, 95% CI 1.38 - 14.12) was associated with LNG-IUS retention.
There are several studies on the factors causing the expulsion of LNG-IUS. For instance, Youm.et.al., showed that distortions of the uterine cavity in women with leiomyoma can complicate the retention of the LNG-IUS and increase the risk of expulsion. A study by Beelen et al found that severe dysmenorrhea is associated with risk of expulsion of LNG-IUS [29]. Another study also showed that LNG-IUS expulsion was not associated with pre-and post- treatment PBAC score and post-treatment VAS score [30]. However, in our study we did not find them as the predictors for retention. A study by Chen et al identified different factors influencing the rate of expulsion in different types of AD categorised based on the MRI findings and found that uterine volume before LNG-IUD insertion (p=0.029, HR: 3.37), menstrual blood loss after Mirena insertion for subtype I , menstrual blood loss after Mirena insertion (p=0.045, HR: 1.02) for intrinsic AD and Intermediate AD respectively [24]. Our study did not gather data on uterine volume and in order to determine the factors associated with LNG-IUS retention, we only examined the AD patients' baseline characteristics.
Limitations of the study
The present study has some limitations. The retrospective nature of the study, and single-centre study presents with inevitable recall bias therefore unknown confounding factors could affect the outcome. Furthermore, this study enrolled patients who had a follow-up of only 6 months, which can be considered short in comparison to other studies which intended to access the treatment success. Failure to capture the adverse events (AE) and failure to collect QOL variables at follow-up also pose limitations in our study.
To validate our results, a large-scale prospective study with an extended follow-up period is necessary.
Future perspective:
Moving forward, advancing the use of LNG-IUS in treating symptomatic AD entails several critical avenues of exploration. First, there's a need to refine patient selection criteria by investigating factors such as specific symptom profiles, uterine characteristics, and responsiveness to initial treatment phases. This approach aims to tailor the use of LNG-IUS more precisely to individual patient needs and optimize treatment outcomes.
Comparative studies are essential to evaluate LNG-IUS effectiveness against alternative treatments like hormonal therapies or surgical interventions over extended periods. These studies should focus on long-term symptom relief, patient satisfaction, and overall treatment efficacy to establish the most suitable therapeutic options for different patient groups.
Extending follow-up periods beyond 60 months is crucial to assess sustained symptom relief, LNG-IUS retention rates, and potential late-onset AE comprehensively. This longer-term perspective will provide valuable insights into the durability and safety of LNG-IUS use in AD management.
Continued monitoring and reporting on the long-term safety profile and tolerability of LNG- IUS are essential. This includes vigilance regarding rare AE and developing appropriate management protocols to ensure patient safety throughout treatment. These initiatives collectively aim to advance the understanding and utilization of LNG-IUS in AD treatment, ultimately improving patient outcomes and QoL in AD patients.
Conclusion
Currently, there are multiple treatment options for patients diagnosed with AD. Hysterectomy being the most well-established treatment with minimal risk of recurrence of disease, another being LNG-IUS. The long-term use of LNG-IUS proves effective and acceptable for managing symptomatic AD. Our study agrees with this and underscores the clinical efficacy and challenges associated with LNG-IUS treatment in AD patients. LNG-IUS which can be used in the treatment of AD either alone or in adjunct with conservative treatments/ other pharmacological therapies. LNG-US significantly alleviates pain related to AD at 1M, 3M & 6 months. Our subgroup analysis of patients treated with LNG-IUS for AD revealed a 6-month retention rate of 68.92%. The factors influencing device retention included baseline symptoms such as dysmenorrhea and dyspareunia.
The present study also demonstrated that LNG-IUS treatment effectively reduces pain associated with AD at 1M, 3M & 6M. We observed significant decreases in pain scores from baseline to 6 months post-insertion, indicating sustained symptom relief over the study period, highlighting the therapeutic benefit of LNG-IUS in managing ADs-related pain.
References
Schrager S, Yogendran L, Marquez CM, Sadowski EA. Adenomyosis: Diagnosis and Management. American Family Physician. 2022 Jan;105(1):33-8.
1. Huang X, Huang Q, Chen S, et al (2015) Efficacy of laparoscopic adenomyomectomy using double-flap method for diffuse uterine adenomyosis. BMC Womens Health 15:. https://doi.org/10.1186/S12905-015-0182-5
2. Y?ld?r?r M, Aytan H, Durukan H, Gürses ? (2022) A clinical scoring system for the diagnosis of adenomyosis. Turk J Obstet Gynecol 19:138–144. https://doi.org/10.4274/TJOD.GALENOS.2022.88289
3. Cai Y, Sun Y, Xu F, et al (2023) Effects of high-intensity focused ultrasound combined with levonorgestrel-releasing intrauterine system on patients with adenomyosis. Scientific Reports 2023 13:1 13:1–10. https://doi.org/10.1038/s41598-023-37096-y
4. Guo S-W (2022) Cracking the enigma of adenomyosis: an update on its pathogenesis and pathophysiology. Reproduction 164:R101–R121. https://doi.org/10.1530/REP-22-0224
5. Harada T, Khine YM, Kaponis A, et al (2016) The Impact of Adenomyosis on Women’s Fertility. Obstet Gynecol Surv 71:557–568. https://doi.org/10.1097/OGX.0000000000000346
6. Moawad G, Kheil MH, Ayoubi JM, et al (2022) Adenomyosis and infertility. J Assist Reprod Genet 39:1027–1031. https://doi.org/10.1007/S10815-022-02476-2
7. Bourdon M, Santulli P, Marcellin L, et al (2021) Adenomyosis: An update regarding its diagnosis and clinical features. J Gynecol Obstet Hum Reprod 50:102228. https://doi.org/10.1016/J.JOGOH.2021.102228
8. Tang Y, Ming-tao Y, Xiang R mei, et al (2022) Preoperative CA125 as a risk factor for symptom recurrence of adenomyosis after ultrasound-guided high-intensity focused ultrasound ablation surgery. Int J Hyperthermia 39:1164–1169. https://doi.org/10.1080/02656736.2022.2107716
9. Özçelik KÖ, Çapar MÇ, Uçar MGU, et al (2016) Are cytokine levels in serum, endometrial tissue, and peritoneal fluid a promising predictor to diagnosis of endometriosis-adenomyosis? Clin Exp Obstet Gynecol 43:569–572. https://doi.org/10.12891/ceog3022.2016
10. Borisov E, Knyazeva M, Novak V, et al (2020) Analysis of Reciprocally Dysregulated miRNAs in Eutopic Endometrium Is a Promising Approach for Low Invasive Diagnostics of Adenomyosis. Diagnostics 10:. https://doi.org/10.3390/DIAGNOSTICS10100782
11. Bourdon M, Santulli P, Kateb F, et al (2021) Adenomyosis is associated with specific proton nuclear magnetic resonance (1H-NMR) serum metabolic profiles. Fertil Steril 116:243–254. https://doi.org/10.1016/J.FERTNSTERT.2021.02.031
12. Yang H, Lau WB, Lau B, et al (2017) A mass spectrometric insight into the origins of benign gynecological disorders. Mass Spectrom Rev 36:450–470. https://doi.org/10.1002/MAS.21484
13. Costanzi F, De Marco MP, Colombrino C, et al (2021) The treatment with Levonorgestrel Releasing Intrauterine System (LNG-IUS) in patients affected by menometrorrhagia, dysmenorrhea and adenomimyois: clinical and ultrasonographic reports. Eur Rev Med Pharmacol Sci 25:3432–3439. https://doi.org/10.26355/EURREV_202105_25823
14. Dai Y, Leng J (2021) The Surgical Treatment of Adenomyosis. Adenomyosis: Facts and treatments 99–111. https://doi.org/10.1007/978-981-33-4095-4_11
15. Madueke-Laveaux OS, Elsharoud A, Al-Hendy A (2021) What We Know about the Long-Term Risks of Hysterectomy for Benign Indication-A Systematic Review. J Clin Med 10:. https://doi.org/10.3390/JCM10225335
16. Abbas AM, Samy A, Atwa K, et al (2020) The role of levonorgestrel intra-uterine system in the management of adenomyosis: A systematic review and meta-analysis of prospective studies. Acta Obstet Gynecol Scand 99:571–581. https://doi.org/10.1111/AOGS.13798
17. Vannuccini S, Petraglia F (2019) Recent advances in understanding and managing adenomyosis. F1000Res 8:. https://doi.org/10.12688/F1000RESEARCH.17242.1
18. Sharara FI, Kheil MH, Feki A, et al (2021) Current and Prospective Treatment of Adenomyosis. J Clin Med 10:. https://doi.org/10.3390/JCM10153410
19. Ota I, Taniguchi F, Ota Y, et al (2021) A controlled clinical trial comparing potent progestins, LNG-IUS and dienogest, for the treatment of women with adenomyosis. Reprod Med Biol 20:427–434. https://doi.org/10.1002/RMB2.12408
20. Li L, Leng J, Jia S, Lang J (2019) Treatment of symptomatic adenomyosis with the levonorgestrel-releasing intrauterine system. Int J Gynaecol Obstet 146:357–363. https://doi.org/10.1002/IJGO.12887
21. Oliveira MAP, Crispi CP, Brollo LC, De Wilde RL (2018) Surgery in adenomyosis. Arch Gynecol
Obstet 297:581–589. https://doi.org/10.1007/S00404-017-4603-6
22. Beatty MN, Blumenthal PD (2009) The levonorgestrel-releasing intrauterine system: Safety, efficacy, and patient acceptability. Ther Clin Risk Manag 5:561–574. https://doi.org/10.2147/TCRM.S5624
23. Sun F, Zhang Y, You M, et al (2021) Laparoscopic adenomyomectomy combined with levonorgestrel-releasing intrauterine system in the treatment of adenomyosis: Feasibility and effectiveness. J Obstet Gynaecol Res 47:613–620. https://doi.org/10.1111/JOG.14571
24. Chen S, Wang J, Sun W, et al (2020) Efficacy of the levonorgestrel-releasing intrauterine device is associated with different subtypes of adenomyosis: a retrospective study. Ann Transl Med 8:1356–1356. https://doi.org/10.21037/ATM-20-3420
25. Yang H, Wang S, Fu X, et al (2022) Effect of modified levonorgestrel-releasing intrauterine system in human adenomyosis with heavy menstrual bleeding. J Obstet Gynaecol Res 48:161. https://doi.org/10.1111/JOG.15031
26. Shaaban OM, Ali MK, Sabra AMA, Abd El Aal DEM (2015) Levonorgestrel-releasing intrauterine system versus a low-dose combined oral contraceptive for treatment of adenomyotic uteri: a randomized clinical trial. Contraception 92:301–307. https://doi.org/10.1016/J.CONTRACEPTION.2015.05.015
27. Radzinsky VE, Khamoshina MB, Nosenko EN, et al (2016) Treatment strategies for pelvic pain associated with adenomyosis. Gynecol Endocrinol 32:19–22. https://doi.org/10.1080/09513590.2016.1232673
28. Youm J, Lee HJ, Kim SK, et al (2014) Factors affecting the spontaneous expulsion of the levonorgestrel-releasing intrauterine system. Int J Gynaecol Obstet 126:165–169. https://doi.org/10.1016/J.IJGO.2014.02.017
29. Beelen P, van den Brink MJ, Herman MC, et al (2021) Predictive factors for failure of the levonorgestrel releasing intrauterine system in women with heavy menstrual bleeding. BMC Womens Health 21:. https://doi.org/10.1186/S12905-021-01210-X
30. Zhang L, Yang H, Zhang X, Chen Z (2019) [Efficacy and adverse effects of levonorgestrel- releasing intrauterine system in treatment of adenomyosis]. Zhejiang Da Xue Xue Bao Yi Xue Ban 48:130–135. https://doi.org/10.3785/J.ISSN.1008-9292.2019.04.02.
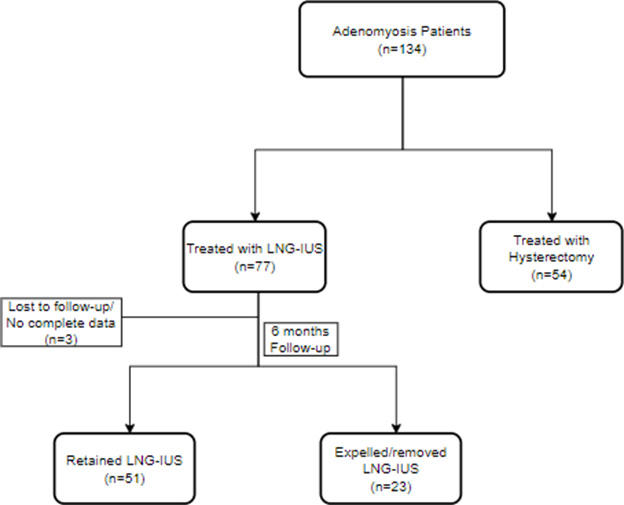
Figure 1
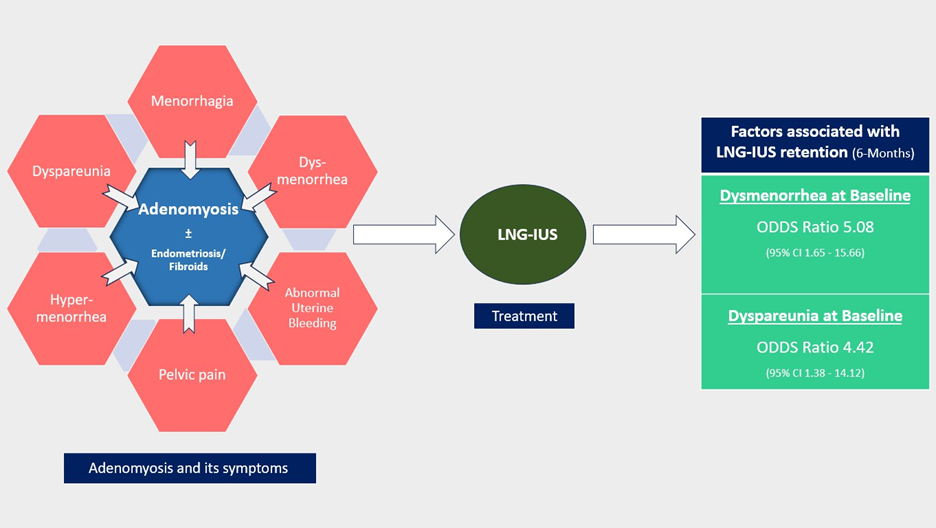
Figure 2
