A Case Report: New Onset Aphasia, Visual Field Defects, and Right-sided Weakness following Semaglutide Administration.
A Case Report: New Onset Aphasia, Visual Field Defects, and Right-sided Weakness following Semaglutide Administration.
Eunheh Koh, MPH1*; Ovais Inamullah, MD2; Alexander Liaugminas, MD2
1. Medical College of Georgia, Augusta, Georgia, United States of America
2. Wellstar Kennestone Hospital, Department of Neurology, Atlanta, Georgia, United States of America.
3.Wellstar Kennestone Hospital, Department of Physical Medicine and Rehabilitation, Atlanta, Georgia, United States of America.
*Correspondence to: Eunheh Koh, MPH, Medical Student at the Medical College of Georgia.
Copyright
© 2025 Eunheh Koh, This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 06 Oct 2025
Published: 15 Oct 2025
Abstract
Background: Embolic strokes of undetermined source (ESUS) are a subtype of cryptogenic stroke that are defined by four primary criteria (signs of ischemia on imaging, absence of cerebral atherosclerotic disease, absence of cardiac abnormalities, and no other identified source of stroke).
Case Presentation: In this case report, we present a 50-year-old female with new onset aphasia, visual field deficits, and mild right-sided weakness secondary to an ischemic stroke, which started one week after her second dose of semaglutide. Her MRI revealed an acute left occipitotemporal infarct in addition to subacute infarcts in the right frontal lobe. She had a negative initial workup, including her cardiology studies, CT angiography and laboratory studies; as a result, her stroke was determined to be cryptogenic in nature. She was discharged on a 21-day course of dual antiplatelet therapy.
Conclusions: The cause of this patient's stroke is yet to be determined, but the temporal relationship between her semaglutide use and symptom onset raises cause for concern. While she lacked cardiovascular risk factors, she was concurrently taking progesterone, which may have further contributed to her risk.
Keywords: embolic stroke of undetermined source (ESUS), semaglutide.
A Case Report: New Onset Aphasia, Visual Field Defects, and Right-sided Weakness following Semaglutide Administration.
Background
Stroke describes a syndrome in which an acute, neurological deficit arises after a hemorrhagic or ischemic injury to the brain, thereby preventing adequate perfusion.1 Ischemic strokes may arise from atherosclerotic disease, cardioembolism, or small vessel infarctions (lacunar strokes).1
Ischemic, non-lacunar strokes with an unknown, but suspected embolic origin are classified as embolic strokes of undetermined source (ESUS).2 This category of stroke comprises 17% of all subtypes of ischemic strokes and 80-90% of all cryptogenic strokes.2,3 Following a standard evaluation, ESUS is diagnosed if the patient meets four clinical criteria. These criteria include: (i) signs of a non-lacunar ischemic stroke on imaging, (ii) absence of atherosclerotic disease in cerebral vasculature, (iii)No cardiac source (including structural abnormalities and arrhythmias), and (iv) no other identified source of stroke.2 The recurrence rate of ESUS is estimated to be 4.5% per year, although this may significantly vary based on age and other patient comorbidities.2 Treatment of ESUS primarily involves antiplatelet therapy with occasional use of adjunctive oral anticoagulant therapy.2
In this study, we describe a case of ESUS in a 50 year old female, which promptly followed the patient’s use of semaglutide for weight loss. Semaglutide is a glucagon-like peptide 1 (GLP-1) agonist that modulates the gut-brain axis and interactions with leptin.4 Its efficacy in weight loss has led to widespread use nationwide.5 The temporal relationship between the medication use and the patient's onset of her stroke raises a concern about potential side effects from GLP-1 agonists.
Case Presentation
A 50-year-old female with a past medical history of endometrial polyps on norethindrone therapy presented to the emergency department with aphasia, right-sided weakness, and vision changes. One week prior to admission, she had received her second injection of semaglutide for weight loss, after which she had fluctuating episodes of aphasia that spontaneously resolved. She decided to come into the hospital due to the presence of a new onset visual field defect upon awakening. Her initial neurological examination revealed expressive and receptive aphasia, a right inferior quadrantanopia, and mild right-sided weakness with a documented strength of 4/5 in both her upper and lower extremities. Pertinent negatives included intact sensation and coordination functions. Her National Institutes of Health Stroke Scale score was a 2.
The only medical history that she reported was abnormal uterine bleeding, for which she was following with her gynecologist. Her obstetrics history was significant for five previous pregnancies, including a history of a miscarriage in her twenties and one induced abortion. She was postmenopausal and upon further investigation, she was diagnosed with two endometrial polyps (one 1.35 cm posterior polyp and one anterior polyp 2.3 x 1.8 cm). She had started progesterone therapy one month prior and was planning to have an elective polypectomy the week of her admission as a possible curative treatment for the bleeding. Of note, her body mass index (BMI) on admission was 24.2. Patient denied any tobacco use and did not have a history of migraine or Sneddon syndrome.
The following differential diagnoses were compiled based on the symptoms of her initial presentation (Table 1). Stroke was on the highest priority amongst the differential diagnoses due to the patient’s acute presentation and unilateral symptoms.
Table 1: Differential diagnoses in Descending Order of Priority
|
Investigative Studies
A non-contrast computed tomography (CT) of the head was negative for an acute intracranial hemorrhage. However, a magnetic resonance image (MRI) revealed an acute left occipitotemporal lobe infarct (Figure 1) in addition to a cluster of subacute infarcts in the lateral right frontal lobe (Figures 2a, 2b and 3). The distribution of these infarcts raised suspicion for an embolic etiology.
Figures 1, 2, and 3 are axial diffusion-weighted images (DWI) from the patient’s MRI. Figure 1 demonstrates the new left occipitotemporal infarct, while Figures 2a and 2b show the subacute infarcts in the lateral right frontal lobe. Figure 3 presents the DWI and FLAIR images for the subacute infarcts in the right frontal lobe.
A thrombolytic intervention was deferred due to the timing of her presentation (with a last known normal greater than four and a half hours6) and the lack of evidence of a large proximal or distal vessel occlusion on CT angiography. The CT angiography was also negative for intracranial atherosclerosis and evidence of carotid disease. She was started on a 21-day course of 75 milligrams of clopidogrel and 325 milligrams of aspirin.
To investigate the possibility of an embolic source, a thorough cardiac workup was completed. There were no abnormal arrhythmias noted on her implantable loop recorder for 10 months. Furthermore, her transthoracic and transesophageal echocardiogram with bubble studies did not reveal a right-to-left shunt or an intracardiac thrombus. These findings suggested that the presence of a cardioembolic source was less likely.7 Her lipid panel was significant for a low high-density lipoprotein of 33 mg/dL (normal: >40 mg/dL) with all other values being within normal limits (Supplemental Table 1). Her thyroid stimulating hormone (1.63 mIU/L) and hemoglobin A1C (5.2%) were normal. Further laboratory studies revealed a slightly decreased partial thromboplastin time of 21 seconds (normal: 25-35 seconds) with a normal international normalized ratio (INR) and prothrombin time (Supplemental Table 1). Her D-dimer was significantly elevated at 1983 ng/mL (normal: <500 ng/mL), which was likely secondary to the acute phase of her stroke.8 A bilateral ultrasound with Doppler was completed on the lower extremities and did not show any sonographic evidence of a deep vein thrombosis (DVT).
Following her hospitalization, she completed an outpatient hypercoagulable workup a few weeks after her stroke, which was significant for slightly decreased protein S activity at 55% (normal: 60-140%; Supplemental Table 1). She also was enrolled in a neurorehabilitation program to guide her recovery. She did not receive a further workup for malignancy due to the lack of other symptoms.
Discussion
In her case, the potential embolism source was not identified, and the case was considered to be cryptogenic following the standard evaluation. In this scenario, the patient is recommended to proceed with an advanced evaluation2,3 – which includes prolonged cardiac rhythm monitoring and hypercoagulability testing - to further delineate a potential cause.
Common etiologies of cryptogenic stroke have been outlined in Table 2.
Table 2: Etiologies of Cryptogenic Stroke2,3,7
|
In the advanced evaluation, her initial hypercoagulable labs were significant for a mild protein S deficiency (Table 2). Protein S is an anticoagulant that inhibits Factors Xa, FVa and FIXa,9 and thus, its deficiency is associated with a prothrombotic state.10 However, there is conflicting evidence in the literature regarding the risk of ischemic stroke in a patient with protein S deficiency. While there have been a few case studies that have elucidated a potential relationship between protein S deficiency and ischemic stroke,11,12 a case-control study failed to determine any significance of protein S deficiency between cases of ischemic stroke and controls.13 In her case, repeat labs may help to assess if she has a true genetic or an acquired deficiency, as protein S levels have been found to fluctuate with various factors such as age and gender.9 Furthermore, in a study completed in 1998, repeat laboratory studies revealed that only 2 out of 20 results were consistent with a true inherited protein S deficiency,14 making it difficult to characterize the impact of this finding. In addition, there were no available medical records that delineated the cause of her previous miscarriage, although the lack of recurrent pregnancy loss (two or more miscarriages15) and three successful pregnancies do not support the presence of an underlying hypercoagulable condition.
However, the patient’s medication history may be of concern in regard to potential sources of embolism. Progesterone-only medications have not been associated with an increased risk of ischemic stroke,16 although a recent case-control study found that individuals who used norethindrone acetate were more likely to develop a DVT compared to non-users and may present a higher risk of thrombosis.17 Thus, the patient’s use of progesterone may have contributed to her overall risk.
In addition to the progesterone’s contribution to her overall risk, the temporal relationship between the onset of her symptoms and the use of semaglutide raises concerns. Semaglutide also has several developing theories regarding its thrombotic properties. A recent meta-analysis suggests that there are potential prothrombotic effects with the use of semaglutide with a notable increase in DVT risk of 266%.18 They predicted that this effect may be due to increased blood viscosity secondary to dehydration, as a common side effect of semaglutide is diarrhea.18 While the findings of this meta-analysis demonstrate increased risk of venous thromboses, they do not support the development of arterial thromboses, which may need to be explored further. In a study of human neutrophil cells, semaglutide was found to activate the secretion of gelsolin and thereby protect against thrombosis.19 In consideration of the limited evidence within the literature, it is important to consider a potential interaction between her medications that may have been a source of her emboli.
This patient had received two injections of semaglutide (at an unspecified dose) for weight loss. However, her BMI of 24.2 does not qualify for the standards listed by the Food and Drug Administration. These regulations state that GLP-1 agonists such as semaglutide should only be prescribed for patients with a BMI of 27 with other comorbidities such as hypertension or with a BMI of 30.20 As a result, our patient may have been more susceptible to the effects of semaglutide. Additionally, the patient was not asked regarding any adverse effects to the medication such as diarrhea at the time of symptom onset, which may have occurred and further contributed to her vulnerability.
As semaglutide is a relatively new medication, providers must take greater caution when prescribing it - particularly for individuals using it for weight loss despite not having a recommended BMI - for possible synergistic prothrombotic effects in a patient with underlying risk factors. To our knowledge, this is the first case in the literature of an ischemic stroke that is temporally associated with semaglutide use, which highlights the need for further research to better characterize its possible hypercoagulable effects.
References
1. Murphy SJ, Werring DJ. Stroke: causes and clinical features. Medicine (Abingdon). 2020;48(9):561-566. doi:10.1016/j.mpmed.2020.06.002
2. Hart RG, Catanese L, Perera KS, Ntaios G, Connolly SJ. Embolic stroke of undetermined source. Stroke. 2017 Apr;48(4):867–72. doi:10.1161/strokeaha.116.016414
3. Saver JL. Cryptogenic stroke. New England Journal of Medicine. 2016 May 26;374(21):2065–74. doi:10.1056/nejmcp1503946
4. García-Vega D, Sánchez-López D, Rodríguez-Carnero G, Villar-Taibo R, Viñuela JE, Lestegás-Soto A, et al. Semaglutide modulates prothrombotic and atherosclerotic mechanisms, associated with epicardial fat, neutrophils and endothelial cells network. Cardiovascular Diabetology. 2024 Jan 3;23(1). doi:10.1186/s12933-023-02096-9
5. Harris E. Poll: Roughly 12% of US Adults Have Used a GLP-1 Drug, Even If Unaffordable. JAMA. 2024;332(1):8. doi:10.1001/jama.2024.10333
6. Davis SM, Donnan GA. 4.5 hours: the new time window for tissue plasminogen activator in stroke. Stroke. 2009 Jun;40(6):2266–7. doi:10.1161/strokeaha.108.544171
7. Ibrahim, F., & Murr, N. (2022, October 17). Embolic stroke. StatPearls [Internet]. 1. Ibrahim F. Embolic stroke [Internet]. U.S. National Library of Medicine; 2022 [cited 2024 December 23]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK564351/
8. Ahmad A, Islam Z, Manzoor Ahmad S, Sarfraz Z, Sarfraz A, Felix M, et al. The correlation of d?dimer to stroke diagnosis within 24 Hours: A meta?analysis. Journal of Clinical Laboratory Analysis. 2022 Feb 4;36(3). doi:10.1002/jcla.24271
9. Pilli VS, Plautz W, Majumder R. The journey of protein S from an anticoagulant to a signaling molecule [Internet]. U.S. National Library of Medicine; 2016 [cited 2025 Jan 27]. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC5973875/
10. Gupta A. Protein S deficiency [Internet]. U.S. National Library of Medicine; 2022 [cited 2025 Dec 27]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK544344/
11. Girolami A, Simioni P, Lazzaro AR, Cordiano I. Severe arterial cerebral thrombosis in a patient with protein S deficiency (moderately reduced total and markedly reduced free protein S): A family study [Internet]. Schattauer GmbH; 1989 [cited 2024 Dec 27]. Available from: https://www.thieme-connect.com/products/ejournals/abstract/10.1055/s-0038-1646544
12. Hooda A, Khandelwal P, Saxena P. Protein S deficiency: Recurrent ischemic stroke in young. Annals of Indian Academy of Neurology. 2009;12(3):183. doi:10.4103/0972-2327.56319
13. Mayer SA, Sacco RL, Hurlet-Jensen A, Shi T, Mohr JP. Free protein S deficiency in acute ischemic stroke. A case-control study. Stroke. 1993 Feb;24(2):224–7. doi:10.1161/01.str.24.2.224
14. Munts AG, van Genderen PJ, Dippel DW, van Kooten F, Koudstaal PJ. Coagulation disorders in young adults with acute cerebral ischaemia. Journal of Neurology. 1997 Dec 16;245(1):21–5. doi:10.1007/s004150050169
15. El Hachem H, Crepaux V, May-Panloup P, Descamps P, Legendre G, Bouet P-E. Recurrent pregnancy loss: Current perspectives. International Journal of Women’s Health. 2017 May;Volume 9:331–45. doi:10.2147/ijwh.s100817
16. Demel SL, Kittner S, Ley SH, McDermott M, Rexrode KM. Stroke risk factors unique to women. Stroke. 2018 Mar;49(3):518–23. doi:10.1161/strokeaha.117.018415
17. Cockrum RH, Soo J, Ham SA, Cohen KS, Snow SG. Association of progestogens and venous thromboembolism among women of reproductive age. Obstetrics & Gynecology. 2022 Aug 3;140(3):477–87. doi:10.1097/aog.0000000000004896
18. Yin D-G, Ding L-L, Zhou H-R, Qiu M, Duan X-Y. Comprehensive analysis of the safety of SEMAGLUTIDE in type 2 diabetes: A meta-analysis of the sustain and pioneer trials. Endocrine Journal. 2021;68(6):739–42. doi:10.1507/endocrj.ej21-0129
19. Zheng Z, Zong Y, Ma Y, Tian Y, Pang Y, Zhang C, et al. Glucagon-like peptide-1 receptor: Mechanisms and advances in therapy. Signal Transduction and Targeted Therapy. 2024 Sept 18;9(1). doi:10.1038/s41392-024-01931-z
20. Tzoulis P, Baldeweg SE. Semaglutide for weight loss: unanswered questions. Front Endocrinol (Lausanne). 2024;15:1382814. Published 2024 Jun 5. doi:10.3389/fendo.2024.1382814.
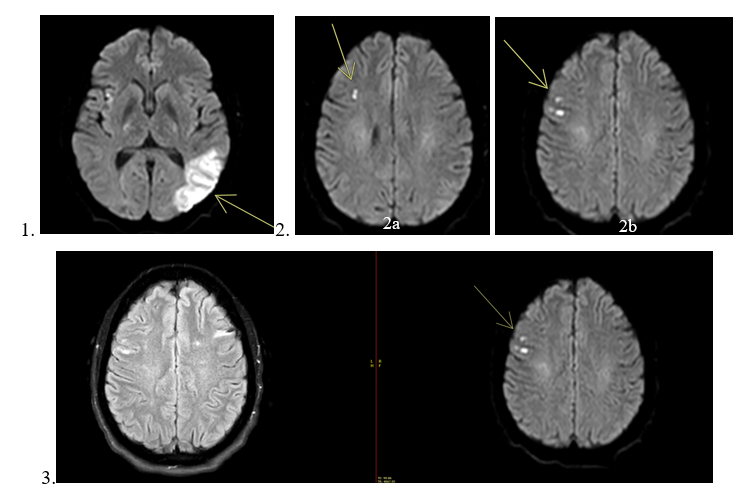
Figure 1
